News
Part of a joint dissemination initiative, RHINOCEROS, along with other members of the Cluster Hub “Materials for Batteries“, took the stage at Battery Innovation Days 2025, contributing to the discussion about the strategic role of recycling in the EU Battery Regulation and the broader circular economy.
Presentations explained, one after the other, Europe’s alternatives to build a circular battery ecosystem. With electric mobility accelerating and battery demand soaring, recycling is no longer optional. It has become a strategic necessity for Europe’s competitiveness and climate goals. And the EU wasted no time to announce on 3 December its recent ReSourceEU Action Plan, under the headline “Accelerating our critical raw materials strategy to adapt to a new reality”. ReSourceEU places circularity at the core of EU’s approach to set the basis for competitive CRMs industry in Europe. Projects similar to RHINOCEROS welcome this initiative and are willing to contribute with expertise and knowledge sourced from R&I.
Take-aways from the BID 2025 sessions
Eleonora Cali (RINA), representing the Materials for Batteries Cluster Hub in the parallel session “End of life, start of supply: Advancing battery recycling in Europe” on 2 December, joined leading experts to address two pressing realities in the battery industry:
- Europe’s dependency on imported raw materials. Lithium, cobalt, nickel, manganese and graphite are critical for the energy transition, yet supply chains remain dominated by non-European players. Recycling offers a way to keep these resources in Europe, reduce environmental impact, and comply with EU regulations on secondary raw material content. The European Commission’s new Battery Regulation aims to change that by mandating minimum recycled content for key materials from 2031. This is more than an environmental measure: it is an industrial policy designed to keep resources within Europe and reduce strategic dependency.
- the expected surge of end-of-life batteries. With EU speeding up its transition to electric mobility, the question of what happens to millions of batteries at the end of their lifetime is shifting from technical to strategic priority.
Surprisingly, speakers underlined EU’s anticipated timeline to develop recycling plants, with a scarce input of end of life applications. According to Andreas Opelt (Saubermacher) and Verena Fuchs (Cylib), for electric vehicle batteries, the timeline for returns is uncertain; early fleets are lasting longer than expected, delaying the recycling ramp-up. Opelt concluded his presentation with a pragmatic message: “The storm of batteries is coming, but if you build capacity too early, plants will sit empty”, arguing timing is critical.
Speakers from both R&I and industry taking the stage in this session called for:
- accelerated permitting for recycling infrastructures. In China, you can build a recycling plant in six months. In Europe, six months is not even enough to submit a permit,” Opel warned.
- enforcement of design-for-recycling standards in new battery regulations.
- support for industrial scale-up through funding but also simplified regulation.
- call to impose all possible measures to prevent black mass from exiting Europe, already reinforced by its recent classification as hazardous waste.
Probably one of the messages we take with us and integrate it to our initiatives’ objectives is that policies like the EU’s Critical Raw Materials Act, Battery Regulation and now recently adopted ReSourceEU provide the framework. What is needed now is execution at speed.

RHINOCEROS project validates extraction routes
The road to a green transition relies heavily on Europe’s capacity to electrify our way out of the use of coal, oil and natural gas before it becomes too late. A large part of this European vision counts on accelerating its transition to electric mobility and therefore upscaling its battery production. With scarce primary resources, EU policy makers push for the recycling of end-of-life [EoL] lithium-ion batteries [LIBs] as a strategic priority. More than recovering the metals trapped in defunct applications, the challenge lies in producing battery-grade materials that can reintegrate the supply chain. The R&I RHINOCEROS project has recently validated its extraction routes at laboratory scale, a preliminary step before pilot-scale implementation.
Electrochemical recovery of lithium
Researchers at Chemistry Department of Sapienza University of Rome [UoS] tested various electrochemical parameters to optimise Li extraction from the black masses [BM] obtained by the mechanical pre-treatment operations conducted in work package [WP4]. Their electrochemical process initially demonstrated its ability to extract Li without causing the dissolution of other cathode elements. Later, the research group replicated the electrochemical conditions in a two-chamber cell, where they achieved 82% Li recovery, later refined to produce LiOH with >99.5%.
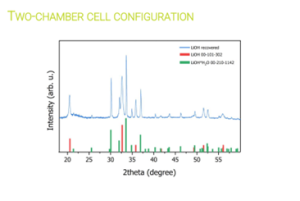
Selective electrochemical recovery of Li using a two-chamber cell configuration. © University of Sapienza
Direct synthesis of cathode and anode materials via hydrometallurgical routes
Additional to electrochemical route, UoS also explored a hydrometallurgical route to synthesise Li-Mn-rich cathodes and reduced graphene oxide (rGO) directly from black mass. Applying Hummers’ method, researchers converted graphite into graphene oxide and co-precipitated metals to form Li-Mn-rich precursors. Electrochemical tests indicate that Li-Mn-rich cathodes derived from thermally treated black mass achieved performance comparable to those made from commercial salts, with capacities up to 202 mAh/g. Reduced graphene oxide synthesised from mechanically treated black mass displayed superior performance compared to rGO from commercial graphite.
Solvometallurgical recovery of Ni and Co
After studying the effects of the pre-treatment processes applied in WP4 to generate BM, researchers at TECNALIA [TEC] validated a solvometallurgical process using deep eutectic solvents (DES) to recover nickel and cobalt under mild conditions. The process achieved >95% metal leaching efficiency and precipitation yields purity.
Moreover, researchers have confirmed the scalability of the solvometallurgical process to large-lab testing without performance loss. To address the costs of this process, TEC researchers have also succeeded in demonstrating the reuse of DES up to 12 times across three systems.
Direct recovery of Ni/Co/Mn/Li with gas-diffusion electrocrystallisation
VITO advanced their proprietary gas-diffusion electrocrystallisation [GDEx] to recover Ni, Co, Mn and Li from leachates obtained from black mass and other WP5 partners. The process delivered >90% recovery rates for Ni, Mn and Co and>99% for Li in the form of layered double hydroxide (LDH) and spinel-type nanostructures for the synthesis of cathode active materials.
With better results obtained from the lithiated nickel manganese cobalt oxide (LNMCO) material synthesised from the leached black mass provided by ACC [thermal pre-treatment], VITO researchers validated its electrochemical activity as a cathode material for LIBs by assembling coin-type half cells. The recycled cathode material showed electrochemical activity, but the achieved capacity is lower than the expected which reveals the requirement of the optimisation process of upscaled extraction of metals, lithiation and slurry processing.
| Sample | Metal recovery (%) | |||
| Ni | Mn | Co | Li | |
| RHINO 2d | 90 | 89 | 96 | 99 |
Optimised recovery of materials from low concentration waste streams
A different task in the refining work package, led by LEITAT and TEC, aims at the recovery of low concentration materials from leachates and effluent streams produced in previous refining tasks. Researchers have developed polymer inclusion membranes (PIMs, LEITAT) and electrochemical systems (TEC) to recover metals from low-concentration streams, achieving up to 80 % recovery for Co and Mn, up to 60 % for Ni and >70 % lithium precipitation with high carbonate purity (>94 %).
In the case of the polymer inclusion membranes, several experiments were performed to define and select both the adequate extractant and the optimal operational conditions. Additionally, selectivity tests are being conducted with a focus on assessing the selectivity of PIM towards a specific metal. Finally, the selected PIMs were defined as follow:
| Series | Thickness | Polymer | Plasticizer | Carrier |
| Mn-PIM-1 | 30±2 um | CTA | 2-NPOE | DEHPA |
| Co-PIM-1 | 28±4 um | CTA | 2-NPOE | Cyanex 272+TBP |
| Ni-PIM-LIX 1 | 40±4 um | CTA | 2-NPOE | LIX84I |
Lab scale validation of most promising routes
After a multi-criteria assessment considering technical performance, eco-efficiency, and scalability, the TEC solvometallurgical route using ACC thermal black mass was selected for pilot-scale validation. This process demonstrated the best balance of recovery efficiency, cost, and environmental impact, and will serve as the reference flowsheet for WP6 upscaling.
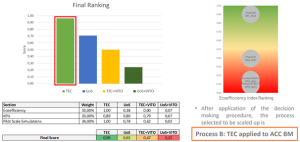
Results evaluation at lab scale and prior to upscaling. ©TECNALIA
On 12 December 2024, the third edition of the annual workshop of the Cluster Hub “Production of Raw Materials for Batteries from European Resources” took place in Brussels, being co-organised by EU-funded projects RHINOCEROS, CRM-geothermal and CICERO. This third edition, along with an increasing number membership, confirm the hub’s role as a dynamic ecosystem that continues to generate innovations in the European battery materials sector.
The hub’s annual workshop, held as a satellite event of the Raw Materials Week 2024, provided once again a platform for presenting the most promising results from participating projects. Two technical sessions covered the entire battery value chain, from raw materials mining to recycling, while the opening conveniently portrayed the policy, the regulatory and strategic frameworks that support and drive the EU R&I initiatives in the battery sector.
Policy perspectives and supporting mechanisms for the battery sector
Susana Xara, Project adviser on raw materials at European Health and Digital Executive Agency (HaDEA), established the discussions tone, navigating through the insights of the Critical Raw Materials Act [CRMA] and the Net Zero Industry Act [NZIA] and focusing on their contribution to securing a sustainable supply of critical raw materials for the European battery industry.
Wouter IJzermans, BEPA Executive Director, presented the long-term vision and potential revisions of their roadmap, emphasising the importance of policy frameworks and incentives in promoting battery innovation and deployment across Europe.
The presentation of Vasileios Rizos from the Centre for European Policy Studies (CEPS) identified various barriers and challenges emerging from the EU policy framework on batteries, based on inputs from 20 companies across the entire battery value chain, including partners from the BATRAW project, member of the Cluster Hub since 2022. The representative of CEPS concluded with a set of policy messages referring to early dialogue channels established between policy-makers and various stakeholders. Before the legal requirements entry into force, this information exchange on availability of secondary data sets could enable stakeholders to assess the data quality, select suitable sets of information and identify potential data gaps.
Publicly available resources submitted by CEPS:
- Barriers and policy challenges in developing circularity approaches in the EU battery sector: an assessment – CEPS
- Implementing the EU digital battery passport – CEPS
- Compliance with the EU’s carbon footprint requirements for electric vehicle batteries – CEPS
Orchestrating the launch and on-going work of the Cluster Hub, PNO Innovation Belgium [part of PNO Group – leader in innovation and funding consultancy], represented by Dr. Nader Akil, concluded the first session with an overview of all EU funding programmes supporting research, innovation and investment in raw materials production for batteries. Additional to the upcoming funding opportunities and guidance on selecting the appropriate funding opportunities based on the status of technology, Dr. Nader Akil introduced another initiative launched by PNO Group – DIAMONDS4IF. This project supports the preparation of Innovation Fund applications, enabling the transfer of H2020 research results into successful ventures and securing investment funding.
Download Funding Schemes presentation
The third session of presentations commenced with an outline of the main findings of the LIFE DRONE project, which concluded in June 2024. Presented by Lorenzo Toro, process engineer at Eco Recycling, the project demonstrated the feasibility of producing high-quality NMC oxide and graphite from recycled batteries. The innovative process confirmed significant environmental benefits, with a reduction of 59 % in terms of kg CO2 eq. Additionally, one plant is estimated to treat 500 tonnes of batteries/year. The technical-economic evaluation of this industrial plant, with a potential capacity of 500 tons/year, showed a return on investment (ROI) of 31.64 % and a payback time (PBT) of 3.16 years. The analysis indicated that attractive payback times could be obtained even with varying prices for NMC and graphite.
Download LIFE DRONE presentation
RHINOCEROS presenting results of the Electrochemical Li recovery strategy from LIBs black mass
The electrochemical Li recovery from Li-ion battery black mass, investigated in the RHINOCEROS project and presented by Prof. Pier Giorgio Schiavi from Univ. of Sapienza reported Li extraction yields from end-of-life (EoL) LIBs in the range of 82 %, and faradaic efficiency compared to commercial cathode materials (close to 100 %). Researchers have investigated potential causes that could explain the relatively low selectivity ranges of Li and other metals available in black mass. The simultaneous oxidation of impurities found in the black mass can be a justified explanation for the preliminary results obtained. Experimental results show that the extraction percentages for Co, Ni, and Mn remain in very low ranges. When contemplating upscaling scenarios, Prof. Schiavi mentioned the researchers are evaluating an alternative approach that enables the treatment of larger quantities of powder without replicating the manufacturing process of LIB electrodes.
Download RHINOCEROS presentation
The session featured also presentations of other initiatives addressing the batteries recycling topic: Benjamin P. Wilson, Senior Scientist, Hydrometallurgy and Corrosion at Aalto University on behalf of the RESPECT project, Miguel Aguilar, researcher at LEITAT Technological Centre for the BATRAW projects, and Joana Gouveia [Researcher at the Institute of Mechanical Engineering and Industrial Management (INEGI) and America Quinteros [Researcher at LUT Univ.] for the ReLiEF initiative.
There is an undeclared competition for better, more efficient batteries which pushes researchers to continue developing new methods for extracting and synthesising electrodic materials.
Recovery of lithium as battery grade material
Lithium (Li) is a key component in batteries and scientists involved in the RHINOCEROS Project have been exploring ways to extract it from recycled materials from used batteries known as “black mass” (BM). But one of the challenges scientists are facing is the reduction of fluoride content in extracted Li. Researchers at KIT tested their mechanochemical process for extracting Li from various BM samples provided by partners ACC and TES. Their experiments engaging reactive milling coupled with various reactive agents aimed to reduce the fluoride content of the aqueous solution. These tests showed that using magnesium as a reactive agent yielded most promising results for Li extraction.
Progress in Lithium-Manganese battery materials and Advancements in reduced graphene oxide production
Research team at Sapienza Univ. of Rome (UoS) has been making progress in developing lithium-manganese-rich materials for battery applications. These materials were produced using an integrated hydrometallurgical process, which includes the production of reduced graphene oxide and a co-precipitation method that leads to the formation of lithium-manganese-rich cathodes. The resulting cathode materials are currently undergoing extensive physicochemical and electrochemical characterizations.
For the synthesis of reduced graphene oxide, the researchers compared two methodologies and observed differences in productivity. These differences are now being investigated to determine whether they are influenced by the thermal pre-treatment of the graphite or by the role of metals present in different oxidation states. The use of mechano-chemically treated powder has demonstrated remarkable productivity, reaching approximately 80%.
Enhanced solvometallurgical processes
As evoked by its name, solvometallurgy uses solvents to extract metals. Researchers at TEC have been optimising the process, using additives as copper (Cu) or hydrogen peroxide (H2O2) when necessary, achieving a high recovery rate of >95%. However, the process also increased the dissolved Cu content, which required additional steps to reduce it. Researchers are now exploring methods like cementation or electrodeposition to recover and reuse the dissolved Cu. The deep eutectic solvents (DES) were already regenerated and reused, result which could bring a positive impact on the process sustainability assessment.
Direct recovery of battery materials
The Gas-Diffusion Electrocrystallisation (GDEx) technology allows the one-step recovery of metals and synthesis of new materials with high added value. In the framework of the RHINOCEROS project, the research team at VITO has been focusing on optimising the GDEx technology to achieve the selective recovery of nickel (Ni), manganese (Mn), and cobalt (Co), contained in leachates from black mass and achieved 90 % extraction of Ni, Mn and Co. This two-step GDEx process facilitated the removal of all the impurities such as Cu, Fe from the leachate solution. Using the GDEx process, VITO researchers have successfully synthesised Layered Double Hydroxide (LDH) materials and spinel-type nanostructures from the synthetic solutions. The LDH materials were lithiated and LiNi0.8Mn0.1Co0.1O2 (LNMCO811) was synthesised, which could be used as a cathode active material for lithium-ion batteries (LiBs). The results obtained with synthetic solutions portray the potential of the method to obtain relevant active cathode materials out of leachate solutions.
Recovery of materials from low concentration waste streams
Aiming towards a zero-waste strategy for the recovery of metals from battery refining waste waters, LEITAT is working on the development and evaluation of novel polymer inclusion membranes (PIM). PIMs are a type of liquid membrane in which the liquid phase, the extractant, is held within a polymeric network. The interest in these membranes has been growing exponentially over the past few years as an alternative separation technique to conventional solvent extraction.
During the previous six months, the team at LEITAT have been investigating two types of membranes that have shown high selectivity, recovering manganese (Mn) and cobalt (Co) from mixed metal solutions. In their future research, LEITAT will use these membranes in combination to ensure increased selectivity of targeted metals.
Optimising lithium carbonate recovery
Lithium carbonate(Li₂CO₃) is another critical material for batteries, and researchers at TEC and LEITAT are working to optimise its recovery from various solutions. This involves fine-tuning the conditions for Li₂CO₃ precipitation, including the influence of pH and the presence of other cations. Tests are currently conducted with both synthetic solutions and real leachates to ensure the effectiveness of the process. Additionally, efforts are underway to automate the recovery process, which includes assembling elements for pH monitoring and CO2 bubbling systems.
Bringing innovations to market
To bring these innovations to market, researchers are preparing to scale up their processes. This involves a selective process based on data collection, life cycle assessment (LCA) and life cycle cost (LCC) analysis, to ensure the best technological routes are chosen to be further upscaled and meet the production demands.
Authors: CHA and PNO
An important component of a Li-ion battery (LiB), the electrolyte has a crucial role in the cell performance. The nonaqueous electrolyte is a multicomponent system consisting of a conductive salt, mainly LiPF6 (Lithium hexafluorophosphate – inorganic component), organic carbonate solvents and additives.
Numerous research initiatives are dedicated to the design of the electrolyte composition, aiming to optimized performance, increased safety, lifetime and streamlined costs. However, once a LiB reaches its end-of-life (EoL) stages, the electrolyte receives less attention in the favour of the valuable metals that can be recycled from the cathode: Li, Co, Mn, Ni, Al, Cu. Moreover, due to its volatility, toxicity and high flammability, the electrolyte recycling is less studied. When ignored, the spent electrolyte reacts with water to form fluoride, leading to its uncontrolled decomposition and/or evaporation, and thus generating an imminent environmental risk. Due to its composition that includes organic solvents, the presence of residual electrolyte in the black mass is considered hazardous, and it is often a reason hampering the recycling process.
Researchers at Chalmers University (CHA) ) developed a sub- and supercritical carbon dioxide (sc-CO2) extraction process to selectively recover the electrolyte from spent LiBs. Sc-CO2 extraction technologies are already widely employed in the food, beverage, pharmaceutical, and cosmetic industry. In a nutshell, sc-CO2 forms when CO2 surpasses its critical point at 31°C and 73.8 bar pressure. In this so-called supercritical state, CO2 demonstrates optimal mass-transfer characteristics and can be fine-tuned to alter its physicochemical characteristics by adjusting pressure and/or temperature. However, CO2 proves ineffective as a solvent for high molecular weight polymers and highly polar ionic compounds. However, the addition of a co-solvent or a modifier can significantly improve the solubility properties of sc-CO2, making this alternative a suitable process to selectively extract LiPF6 after solvent removal.
Researchers at CHA investigated different process parameters (pressure, temperature, extraction times) to understand their impact on the extraction behavior of different electrolyte solvents such as ethylene carbonate. Their analysis included both qualitative and quantitative results indicating the composition and the purity. Simultaneously, the research also monitored the process exhaust stream for potential formation of LiPF6 decomposition products. The findings showed that that the non-polar electrolyte solvents like dimethyl carbonate, diethyl carbonate, and ethyl methyl carbonate were successfully extracted using low-density CO2. The more polar electrolyte components such as ethylene carbonate and propylene carbonate were stepwise extracted by gradually increasing the system’s pressure. The developed technology is a game changer not only for electrolyte recycling but also for increased workplace and transportation safety due to removal of the flammable and hazardous substances from battery black mass. The simplicity of the processing design is an additional advantage of the technology.
© Photo credit: Adobe
During the past years, the increase in the use of lithium-ion batteries (LIBs) has become more prominent. An unsurprising trend, whatsoever, due to the widespread and rapid adoption of clean mobility applications, electronic devices, and energy storage systems. Despite their undeniable environmental and social benefits, several challenges lie ahead. In 2022, global lithium demand exceeded the supply despite an 180% increase, IEA reports. Recent communications already forecast the demand of lithium (Li) is expected to soar over the next decade, with mobility accounting for the main consuming market.
The current supply of primary resources is deemed insufficient for the growing demand. Approximately 60% of today’s lithium is mined for battery-related applications, a figure that could reach 95 percent by 2030, McKinsey reports estimate. But this rapid increase in the use of LIBs in EVs will introduce a large quantity of spent batteries in the near future. The alternatives to manage spent batteries include remanufacturing, repurposing and recycling, with the latter one playing a significant role from both ecologic and economic points of view.
Current recycling processes lack selectivity in recovery control and require significant consumption of reagents and energy. The research conducted by the RHINOCEROS partners at Sapienza University of Rome (UoS), Department of Chemistry, aims to develop an electrochemical process for selective extraction of Li from electrodic powder of end-of-life (EoL) LIBs. This concept simulates the charging process of a LIB with an aqueous electrolyte and a cathode material (counter electrode) that facilitates water reduction. The hydroxyls freed by water reduction and the Li + cations deintercalated by the anode will form a LiOH solution.
Using two samples – a commercial powder, respectively one coming from EoL LIBs, and testing the delithiation process using various parametres, UoS researchers obtained:
- Li extraction of 99% from commercial powder
- Li extraction of 82% from waste powder
The lower Li extraction on EoL electrode powders compared to commercial ones is due to the presence of SuperP [a high purity and structured carbon black powder with a moderate surface area for the lithium-ion industry], which oxidises under delithiation conditions.
Discover the scientific publication
RHINOCEROS attending Shifting Economy Week
From 21 to 25 November 2023, the city of Brussels hosted the Shifting Economy Week, an annual event dedicated to showcasing transformative projects that aim to pave the way to an economy that is low-carbon, regenerative, and equally circular. The 2023 exhibitors’ line-up included, among other regional stakeholders, our partner Watt4Ever (W4E), industrial partner specialised in the development of innovative solutions for energy storage and management. W4E leveraged its presence at Shifting Economy Week to to raise awareness about the importance of circular economy principles in the context of the battery industry.
During the same event, W4E’s CEO, Aimilios Orfanos, was invited to speak at the BeCircular conference, an event dedicated to presenting concrete examples of circular economy approaches put in place by Brussels-based companies. He shared insights from W4E’s experience in developing second-life battery systems for electric vehicles, emphasising their potential benefits in terms of environmental impact and cost savings. Simultaneously, the CEO also highlighted the challenges faced by the industry in implementing circular business models, including regulatory barriers and market incentives.

RHINOCEROS at its second participation at Circular Wallonia Days
A few days after attending Shifting Economy Week, W4E represented the RHINOCEROS project at the Circular Wallonia Days, held on 13 and 14 December 2023. Centred around advancing the circularity of the batteries value chain, the event brought together stakeholders from academia, industry, and government to discuss strategies for improving the sustainability of battery production, use, and disposal. The focus topics covered recycling technologies, supply chain transparency, and policy measures to support the transition to a circular battery economy.
© Photo credits: Watt4Ever
Despite a different objective, the RHINOCEROS project partners have shown growing interest in the Digital Battery Passport, an initiative of FREE4LIB, a sister project from the Cluster Hub “Production of raw materials for batteries from European resources”. This collaboration shows our commitment to contributing to the European battery community through the exchange of knowledge and experience.
The FREE4LIB workshop had a three-fold objective, including a brief presentation of the preliminary results of the battery passport concept development, the outline of the implementation challenges and potential follow-ups of industrial scale-up, and the clear differentiation between battery second use (B2U) versus recycling. The event drew approximately 50 participants from various segments of the battery value chain, which ensured a comprehensive and multifaceted perspective of the subject matter.
The introductive session presented the FREE4LIB project, briefly highlighting past achievements and focusing mainly on the remaining activities outlined in the workplan. The following session was led by Julius Ott (industrial engineer with expertise in circular economy at Karl-Franzens-Universität Graz). During the past year, researchers at Univ. of Graz worked on finalising data collection and processing related to the development of a data model of the digital passport platform which aims to close the information gap between beginning-of-life (BoL) and end-of-life (EoL) battery lifetime. This interactive session turned out to be an appropriate opportunity for researchers at Univ. of Graz to present the outcomes of their data collection and handling, and to evaluate their relevance within the reality portrayed by the workshop attendees.
Participants, predominantly familiarised with the EU-funded battery projects, confirmed the findings reported by Univ. of Graz. However, they also raised concerns about data sharing. The outcomes of the interactive session, complementing prior research, will serve as valuable guidance for the FREE4LIB project in implementing the battery passport within their project.
For additional background information on the digital passport developed by FREE4LIB, please refer to previous articles.
Part of the Cluster Hub “Production of raw materials for batteries from European resources”, the RHINOCEROS consortium received the online visit of FREE4LIB representatives during the second day of the Consortium meeting held in Gothenburg. This initiative including stakeholders involved in different European R&D initiatives goes beyond building a knowledge exchange ecosystem to address common topics related to EU-funded projects; it paves new collaboration routes and synergies aiming at driving innovations for the recycling of batteries and the production of raw materials for battery applications from primary and secondary resources available in Europe.
Represented by Julius Ott (industrial engineer with expertise in circular economy at Karl-Franzens-Universität Graz) and Pau Sanchis (senior policy officer Eurobat), the FREE4LIB presentation focused mainly on the Digital Battery passport and the relevant legislative situation at European level.
Pau Sanchis referred to the Digital Battery Passport in the context of the new regulation on batteries and waste batteries which entered into force on 17 August 2023. According to this update, thoroughly explained in Art. 77, the battery passport should contain information “relating to the battery model and information specific to individual battery, including resulting from the use of that battery”.
“Batteries should be labelled in order to provide end-users with transparent, reliable and clear information about batteries and waste batteries. That information would enable end-users to make informed decisions when buying and discarding batteries and waste operators to appropriately treat waste batteries. Batteries should be labelled with all the necessary information concerning their main characteristics, including their capacity and the amount of certain hazardous substances present. To ensure the availability of information over time, that information should also be made available by means of QR codes which are printed or engraved on batteries or are affixed to the packaging and to the documents accompanying the battery and should respect the guidelines of ISO/IEC Standard 18004:2015. The QR code should give access to a battery’s product passport. Labels and QR codes should be accessible to persons with disabilities, in accordance with Directive (EU) 2019/882 of the European Parliament and of the Council (17).”
The policy officer emphasised the role of the standardisation process on the Battery Passport, which requires the Commission to adopt implementing decision requesting European Standardisation Organisation to develop standards in support of Ecodesign by December 2023. Standards regarding the technical design and operation of the Battery Passport are expected to complement provisions under Art. 78. According to the timeline presented in the regulation, the first application of the battery passport is expected in 2027. From 18 February 2027 onwards, “all batteries shall be marked with a QR code as described in Part C of Annex VI.
The new regulation aiming at strengthening sustainability rules for batteries and waste batteries will be supported by various secondary legislation pieces which will ensure all the requirements will be developed and implemented effectively. The QR code shall provide access to the following:
- for light means of transport (LMT) batteries, industrial batteries with a capacity greater than 2kWh and electric vehicles batteries, the battery passport in accordance with Article 77.
- for other batteries, the applicable information referred to in paragraphs 1 to 5 of this Article, the declaration of conformity referred to in Article 18, the report referred to in Article 52(3) and the information regarding the prevention and management of waste batteries laid down in Article 74(1), points (a) to (f).
- for starting, light, and ignition (SLI) batteries, the amount of cobalt, lead, lithium or nickel recovered from waste and present in active materials in the battery, calculated in accordance with Article 8.
The policy overview presentation was complemented by the technical presentation undertaken by Karl-Franzens-Universität Graz, that will develop a data model of the digital battery passport platform aiming to close the information gap between beginning-of-life (BoL) and end-of-life (EoL) battery lifetime. Relying on knowledge generated previously by Univ. of Graz, the researchers set an objective to define clear user roles and establish access to certain information. Up to this moment, the work carried out has been focusing on data collection and data handling (data points sorting) on the other side. This specific work encountered various challenges, notably the users willingness to share information, process standardisation, the variety of products, the recycling cost/revenue ratio, the dynamic development of the legislative framework, to name a few.
Learn more about the progress on the battery passport on the FREE4LIB website.
During the second semester, researchers from KIT further studied and improved the conditions for the mechanochemical transformation of Black Mass (BM). With the BM supplied by ACC is already in a reduced state, the focus now shifts towards reducing the BM provided by partner TES.
This BM consists mostly of nickel-manganese-cobalt (NMC) cathode material and graphite, and it was found to cause a longer reaction time. It is expected that the graphite exfoliates during milling and creates thin protective layers around NMC particles and the reducing agent, slowing down the reaction kinetics.
Using variations of milling parameters like ball-to-sample ratio or the type and amount of reducing agent, researchers optimised the process towards the fastest kinetics. During the investigation, no intermediate reduction to transition metal oxides in lower oxidation states was observed. However, the full reduction of the respective part to the metallic composite occurred.
The operation took place in a shaker mill, and it requested an excess of 3.3 equivalents of Aluminium (Al) towards NMC was required to attain a complete reduction within a reasonable time. This transformation was accompanied by the formation of aluminium carbide and the presence of residual fine aluminium powder which caused a self-ignition hazard when the milled powder came into contact with air.
In contrast, when using calcium as a reactive agent, researchers observed a fast reaction but they also reported a strong dependency of reaction kinetics on calcium size. The hazard of self-ignition when exposed to air was limited in this case by using stoichiometric amounts.
In addition, the research team has been conducting preliminary experiments for the scale-up process using a planetary mill. It was observed that the choice of the milling type has a significant impact on the reaction kinetics. Attempts to crush calcium pieces proved to be unsuccessful, therefore not initiating any reaction. However, when using aluminium, reactions occurred much faster.
In the months ahead, the research team working in WP4 will continue to investigate the milling parameters for the planetary mill.
Find out more information about the activities developed in WP4 in the article Reactive milling for the production of metallic black mass (MBM) – Rhinoceros project (rhinoceros-project.eu)
Read more about the black mass leaching operations in the article Materials extraction and direct routes for the synthesis of electrode materials: Recovery of Lithium as battery grade materials
