News
Part of a joint dissemination initiative, RHINOCEROS, along with other members of the Cluster Hub “Materials for Batteries“, took the stage at Battery Innovation Days 2025, contributing to the discussion about the strategic role of recycling in the EU Battery Regulation and the broader circular economy.
Presentations explained, one after the other, Europe’s alternatives to build a circular battery ecosystem. With electric mobility accelerating and battery demand soaring, recycling is no longer optional. It has become a strategic necessity for Europe’s competitiveness and climate goals. And the EU wasted no time to announce on 3 December its recent ReSourceEU Action Plan, under the headline “Accelerating our critical raw materials strategy to adapt to a new reality”. ReSourceEU places circularity at the core of EU’s approach to set the basis for competitive CRMs industry in Europe. Projects similar to RHINOCEROS welcome this initiative and are willing to contribute with expertise and knowledge sourced from R&I.
Take-aways from the BID 2025 sessions
Eleonora Cali (RINA), representing the Materials for Batteries Cluster Hub in the parallel session “End of life, start of supply: Advancing battery recycling in Europe” on 2 December, joined leading experts to address two pressing realities in the battery industry:
- Europe’s dependency on imported raw materials. Lithium, cobalt, nickel, manganese and graphite are critical for the energy transition, yet supply chains remain dominated by non-European players. Recycling offers a way to keep these resources in Europe, reduce environmental impact, and comply with EU regulations on secondary raw material content. The European Commission’s new Battery Regulation aims to change that by mandating minimum recycled content for key materials from 2031. This is more than an environmental measure: it is an industrial policy designed to keep resources within Europe and reduce strategic dependency.
- the expected surge of end-of-life batteries. With EU speeding up its transition to electric mobility, the question of what happens to millions of batteries at the end of their lifetime is shifting from technical to strategic priority.
Surprisingly, speakers underlined EU’s anticipated timeline to develop recycling plants, with a scarce input of end of life applications. According to Andreas Opelt (Saubermacher) and Verena Fuchs (Cylib), for electric vehicle batteries, the timeline for returns is uncertain; early fleets are lasting longer than expected, delaying the recycling ramp-up. Opelt concluded his presentation with a pragmatic message: “The storm of batteries is coming, but if you build capacity too early, plants will sit empty”, arguing timing is critical.
Speakers from both R&I and industry taking the stage in this session called for:
- accelerated permitting for recycling infrastructures. In China, you can build a recycling plant in six months. In Europe, six months is not even enough to submit a permit,” Opel warned.
- enforcement of design-for-recycling standards in new battery regulations.
- support for industrial scale-up through funding but also simplified regulation.
- call to impose all possible measures to prevent black mass from exiting Europe, already reinforced by its recent classification as hazardous waste.
Probably one of the messages we take with us and integrate it to our initiatives’ objectives is that policies like the EU’s Critical Raw Materials Act, Battery Regulation and now recently adopted ReSourceEU provide the framework. What is needed now is execution at speed.

RHINOCEROS project validates extraction routes
The road to a green transition relies heavily on Europe’s capacity to electrify our way out of the use of coal, oil and natural gas before it becomes too late. A large part of this European vision counts on accelerating its transition to electric mobility and therefore upscaling its battery production. With scarce primary resources, EU policy makers push for the recycling of end-of-life [EoL] lithium-ion batteries [LIBs] as a strategic priority. More than recovering the metals trapped in defunct applications, the challenge lies in producing battery-grade materials that can reintegrate the supply chain. The R&I RHINOCEROS project has recently validated its extraction routes at laboratory scale, a preliminary step before pilot-scale implementation.
Electrochemical recovery of lithium
Researchers at Chemistry Department of Sapienza University of Rome [UoS] tested various electrochemical parameters to optimise Li extraction from the black masses [BM] obtained by the mechanical pre-treatment operations conducted in work package [WP4]. Their electrochemical process initially demonstrated its ability to extract Li without causing the dissolution of other cathode elements. Later, the research group replicated the electrochemical conditions in a two-chamber cell, where they achieved 82% Li recovery, later refined to produce LiOH with >99.5%.
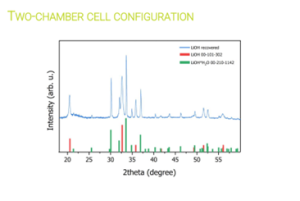
Selective electrochemical recovery of Li using a two-chamber cell configuration. © University of Sapienza
Direct synthesis of cathode and anode materials via hydrometallurgical routes
Additional to electrochemical route, UoS also explored a hydrometallurgical route to synthesise Li-Mn-rich cathodes and reduced graphene oxide (rGO) directly from black mass. Applying Hummers’ method, researchers converted graphite into graphene oxide and co-precipitated metals to form Li-Mn-rich precursors. Electrochemical tests indicate that Li-Mn-rich cathodes derived from thermally treated black mass achieved performance comparable to those made from commercial salts, with capacities up to 202 mAh/g. Reduced graphene oxide synthesised from mechanically treated black mass displayed superior performance compared to rGO from commercial graphite.
Solvometallurgical recovery of Ni and Co
After studying the effects of the pre-treatment processes applied in WP4 to generate BM, researchers at TECNALIA [TEC] validated a solvometallurgical process using deep eutectic solvents (DES) to recover nickel and cobalt under mild conditions. The process achieved >95% metal leaching efficiency and precipitation yields purity.
Moreover, researchers have confirmed the scalability of the solvometallurgical process to large-lab testing without performance loss. To address the costs of this process, TEC researchers have also succeeded in demonstrating the reuse of DES up to 12 times across three systems.
Direct recovery of Ni/Co/Mn/Li with gas-diffusion electrocrystallisation
VITO advanced their proprietary gas-diffusion electrocrystallisation [GDEx] to recover Ni, Co, Mn and Li from leachates obtained from black mass and other WP5 partners. The process delivered >90% recovery rates for Ni, Mn and Co and>99% for Li in the form of layered double hydroxide (LDH) and spinel-type nanostructures for the synthesis of cathode active materials.
With better results obtained from the lithiated nickel manganese cobalt oxide (LNMCO) material synthesised from the leached black mass provided by ACC [thermal pre-treatment], VITO researchers validated its electrochemical activity as a cathode material for LIBs by assembling coin-type half cells. The recycled cathode material showed electrochemical activity, but the achieved capacity is lower than the expected which reveals the requirement of the optimisation process of upscaled extraction of metals, lithiation and slurry processing.
| Sample | Metal recovery (%) | |||
| Ni | Mn | Co | Li | |
| RHINO 2d | 90 | 89 | 96 | 99 |
Optimised recovery of materials from low concentration waste streams
A different task in the refining work package, led by LEITAT and TEC, aims at the recovery of low concentration materials from leachates and effluent streams produced in previous refining tasks. Researchers have developed polymer inclusion membranes (PIMs, LEITAT) and electrochemical systems (TEC) to recover metals from low-concentration streams, achieving up to 80 % recovery for Co and Mn, up to 60 % for Ni and >70 % lithium precipitation with high carbonate purity (>94 %).
In the case of the polymer inclusion membranes, several experiments were performed to define and select both the adequate extractant and the optimal operational conditions. Additionally, selectivity tests are being conducted with a focus on assessing the selectivity of PIM towards a specific metal. Finally, the selected PIMs were defined as follow:
| Series | Thickness | Polymer | Plasticizer | Carrier |
| Mn-PIM-1 | 30±2 um | CTA | 2-NPOE | DEHPA |
| Co-PIM-1 | 28±4 um | CTA | 2-NPOE | Cyanex 272+TBP |
| Ni-PIM-LIX 1 | 40±4 um | CTA | 2-NPOE | LIX84I |
Lab scale validation of most promising routes
After a multi-criteria assessment considering technical performance, eco-efficiency, and scalability, the TEC solvometallurgical route using ACC thermal black mass was selected for pilot-scale validation. This process demonstrated the best balance of recovery efficiency, cost, and environmental impact, and will serve as the reference flowsheet for WP6 upscaling.
Results evaluation at lab scale and prior to upscaling. ©TECNALIA
The last consortium meeting included a session dedicated to ongoing clustering activities, where the RHINOCEROS partners invited the RESTORE project coordinator, Christophe Aucher, Area Manager Energy Storage at LEITAT. The objective of this initiative is to introduce the two EU-funded projects, both focusing on advancing battery material treatment and recycling technologies. Both projects are members of the Cluster Hub “Production of raw materials for batteries from European resources”.
The RESTORE project introduced its approach to treat black mass – from sorting to discharging, retrieving all available materials and validating them in a coin battery. This concept aligns closely with the philosophy of the RHINOCEROS project, emphasising the importance of contactless sorting and efficient battery discharge processes. The aim of RESTORE is to remove cables and discharge batteries within 40 minutes, which would bring a big step towards more efficient recycling processes.
During the event, representatives of both projects addressed various industry challenges. One major issue is the profitability of selling black mass to states outside of EU, compared to the domestic scenario for producing electrodes. Additionally, the European market faces a slowdown in battery production, showing citizens preference for hybrid cars over electric vehicles. Nevertheless, both sides agreed on the need for critical raw materials, indispensable to emerging industrial sectors, underscoring the importance of sustainable and efficient recycling processes.
The RESTORE project introduced several technological advancements, including the KYBURTZ cell-to-electrode direct recycling method developed by VITO. This method aims to streamline the recycling process and enhance the quality of recovered materials.
Although the two projects share similar objectives and consortium partners, technical questions were not missing, addressing mainly the electrochemical recovery of lithium, the impact of the aqueous media on the quality of retrieved material and the potential to work in batch mode.
This first online encounter of RHINOCEROS and RESTORE sparks a lot of curiosity, key ingredient of innovation and technological advancement. Prospects already show opportunities to stimulate collaborations and knowledge exchange in the battery recycling R&I field.
Lithium-ion batteries proved to be one of the most efficient energy storage solutions, widely used for applications such as electric vehicles [EVs] and renewables. Despite faltering economies and material shortages, more than 40 million EVs are expected on the EU’s roads by 2030. With mass electrification of transport, there will be an important demand for batteries and the materials they are made from. Despite an incentivising regulatory context at EU level, in 2023 Europe was home to only 1% of the production of key battery raw materials. European carmakers and battery cell producers are already fret about volatile prices and limited supplies of cathode and anode materials. But with EV fleet number growing on the EU’s roads, the volumes of End-of-life [EoL] batteries will considerably increase in the near future. And so will the pressure to recycle closer to home, recovering key components and mitigating the supply chain risks.
Conventional recycling methods often require high consumption of energy and reagents. But electrochemistry offers a promising green alternative method, enabling selective recovery of Li without the need for chemical extracting compounds.
Selective electrochemical extraction of lithium
Researchers at Chemistry Department of Sapienza University of Rome [UoS] tested various electrochemical parameters to optimise Li extraction from both commercial cathode material and the black masses [BM] obtained by the mechanical pre-treatment operations conducted in work package [WP] 4.
The electrochemical tests on high-purity commercial cathode materials [LiMn2O4, LiCoO2 and LiNi1/3Mn1/3Co1/3O2] allowed a selective extraction of Li up to 98 %. UoS researchers later applied the same electrochemical process on the black mass recovered by the mechanical pre-treatment, obtaining a lower Li extraction yield of 82 %. This decreased yield was mainly attributed to the presence of conductive carbon which undergoes a simultaneous electrochemical oxidation. Analysis of the resulting electrolyte at the end of delithiation experiments showed negligible extraction of cobalt (Co), nickel (Ni), and manganese (Mn). This demonstrated the method’s ability to selectively extract Li without causing the dissolution of other cathode elements.
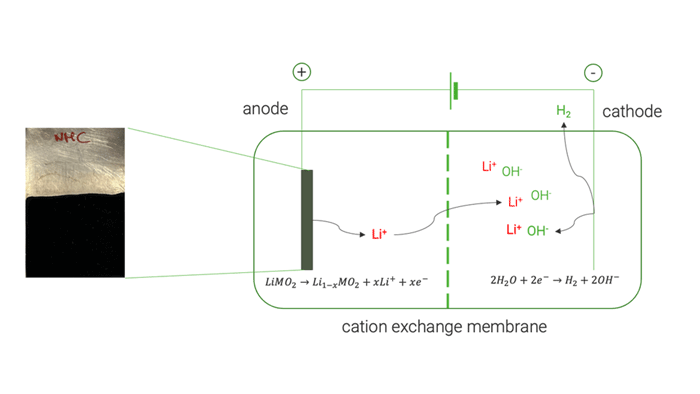
Cation exchange membrane | © Univ. of Sapienza
The optimised electrochemical conditions applied on the BM were replicated in a two-chamber cell to facilitate the separation and concentration of extracted Li on the cathode side, achieving approximately 85 %. The resulting solution was used to crystallise LiOH, resulting in a mixture of LiOH and LiOH·H₂O, with a purity exceeding 99.5 %.
Synthesis of reduced graphene oxide (rGO) and Li-Mn rich cathode material
The direct synthesis of high-value products from EoL LIBs, bypassing the complex and expensive separation of different metals, can be achieved through a competitive recycling strategy. The simultaneous synthesis of reduced graphene oxide (rGO) and lithium-manganese-rich (Li1.2Mn0.55Ni0.15Co0.1O2 – LMR) cathode material from EoL LIBs represents a promising approach to enhance the economic feasibility of hydrometallurgical recycling processes by producing high-value-added advanced materials.
UoS researchers developed an innovative recycling process to directly synthesise a layered LMR cathode and rGO from EoL LIBs. The proposed recycling strategy relies on the application of Hummers’ method to process the electrodic powder delivered by pilot-scale mechanical pre-treatment of mixed Li-ion batteries. The Hummers’ method has allowed for the quantitative extraction of metals from the electrodic powder and the production of GO without any metal impurity. The resulting solution contains the metals from the cathode materials (Co, Ni, Mn) with a significant amount of manganese from the KMnO4 used in the Hummers’ method. From such consideration stems the idea to synthesise LMR cathode material.
UoS researchers synthesised GO using the black masses obtained from three distinct EoL LIB pretreatments performed in WP4. The effects of thermal, mechanical and mechanochemical pretreatments were evaluated using the widely adopted Hummers’ method for graphene production. The study conducted by UoS researchers examined both pristine black masses and those subjected to metal removal via conventional acid leaching, a process frequently employed in LIB recycling to extract metals from cathode materials. This metal removal procedure had a notable effect on introducing oxygen functional group defects across all samples. A synergistic effect was particularly evident in the mechanochemically treated black mass, where acid leaching increased the GO yield significantly. This remarkably high graphite conversion to GO underscores the importance of selecting appropriate pretreatment methods for EoL LIBs, particularly when aiming to integrate advanced materials like GO into the recycling process.
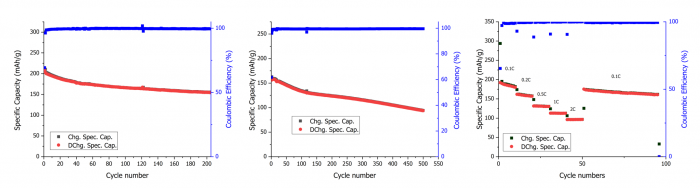
Cycling performance of LMR synthesised from thermally treated black mass: (a) Galvanostatic cycling at 0.1C, (b) Galvanostatic cycling at 1C, and (c) Rate capability performance with the current increasing from 0.1C to 2C every 10 cycles | © UoS
Recovery of Ni/Co materials by solvometallurgical route
RHINOCEROS is a research project that aims to o create economically and environmentally sustainable methods for reusing and recycling LIBs. The project focuses on developing cost-efficient, flexible and eco-friendly processes to recycle all materials in LIBs, including metals, graphite, fluorinated compounds and polymers.
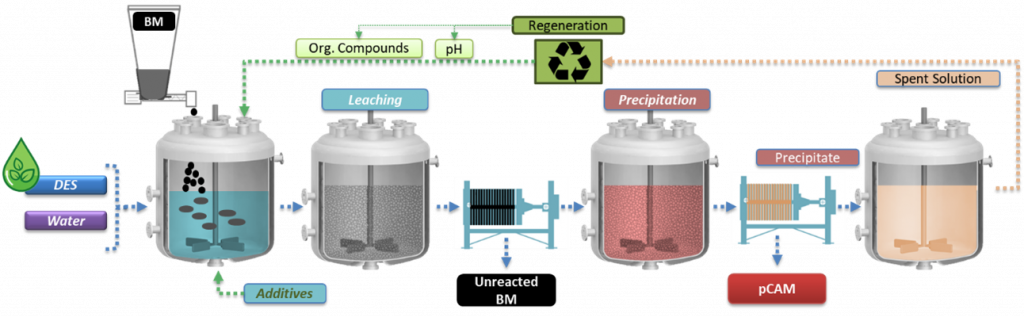
Flowsheet of the leaching, precipitation and regeneration process | © TEC
After studying the effects of the pre-treatment processes applied in WP4 to generate BM, researchers at TECNALIA [TEC] tested various solvometallurgical routes to extract critical metals like Ni, Mn and Co from LIBs under mild conditions. They established optimal process conditions to achieve leaching and precipitation efficiency along with high purity of the produced precursor of cathode active materials (pCAM) under mild conditions. This approach also enhances the recyclability of the extractant.
After characterising all the input materials received from the pretreatment operations, TEC research group analysed also the obtained leachates. The leaching process was optimised at laboratory scale by studying the effects of various parameters such as leaching time and temperature, stirring, waste/liquid ratio and additives.
The solvometallurgical process for recycling end-of-life [EoL] battery BM has successfully developed high-yield, selective systems, validated across various NMC chemistries and black mass pretreatments, achieving over 95 % leaching efficiency. Controlled precipitation delivered up to 95 % precipitation efficiency, producing precursors with more than 99 % purity, meeting project targets. Moreover, researchers have confirmed the scalability of the solvometallurgical process to large-lab testing without performance loss. To address the costs of this process, TEC researchers have also investigated alternatives to reuse and recycle the deep eutectic solvent [DES] leaching spent solution, reporting its dependence on the properties of the BM feed.
The residue after leaching – the graphitic anode material – has been purified via hydrometallurgical routes, underscoring the difficulty of removing the most refractory phases resistant to harsh leaching conditions.
Optimised recovery of materials from low concentration waste streams
A different task in the refining work package, led by LEITAT and TEC, aims at the recovery of low concentration materials from leachates and effluent streams produced in previous refining tasks. It is divided in three subtasks, each exploring different membrane-based technologies to achieve a zero-waste strategy for recovering remaining elements.
The objective of the first subtask is to develop new extractants based on ionic liquids or DESs, to be used within novel Polymer Inclusion Membranes (PIMs), for the recovery of Li from solvometallurgical refining final streams. The team firstly analysed the SoA on Li extraction strategies, selected, synthesised and characterised different potential extractants based on hydrophobic deep eutectic solvents (HDESs). Selected HDESs were synthesised and sent to the next subtask devoted to PIM fabrication and testing using different ILs and/or synthesized HDESs. These membranes represent an innovative technology that effectively adapts to the specificities of each input stream, enabling optimal recovery of valuable metals present in the waste generated by processes carried out. In the this subtask, the PIMs yielded the following recovery rates:
- Co and Mn: efficiencies close to 80 %
- Ni: 60 % recovery
- Li: only 19 %, but exhibiting a high flux rate of 1.2 mol/m²·h, suggesting potential for larger lithium recovery.
Finally, in the last subtask, researchers developed a three-chamber electrochemical process for the extraction, concentration and recovery of lithium in the form of lithium carbonate from solvometallurgical final streams. Different ion-exchange membranes were characterised and lithium carbonate precipitation step was studied and optimised. They tuned the overall process conditions achieving more than 70 % Li extraction from leachate to catholyte and high lithium carbonate precipitation yields (>70 %) with high purity (>94 %).

Three-chamber electrochemical cell | © TEC
Extraction of Ni/Co/Mn/Li with GDEx
Gas-diffusion electrocrystallisation [GDEx] is an innovative electrochemical process used to recover metals from complex fluids, such as leachates, industrial waste streams and organic solutions. This innovative process has been successfully demonstrated in multiple research projects, proving its capability to recover valuable metals while operating under low-cost and environmentally friendly operating conditions. The GDEx process has achieved Technology Readiness Level [TRL] 3 for the recovery of Co and Mn from synthetic solutions and attained TRL 4 in treating black mass leachates. One of the multiple advantages of this electrochemical process is its capacity to recover Ni, Co and Mn simultaneously, with the flexibility to manipulate operational conditions to obtain targeted products.
Specifically for the battery recycling sector, the GDEx process offers various advantages over conventional battery recycling methods:
- High recovery efficiencies, exceeding 90 % for Co and Mn and over 90 % for Li, with potential for further optimisation.
- Low energy consumption: less than 20 kWh per kg of recovered material, which reduces the operational costs significantly.
- Minimal chemical input: the process relies primarily on air and in-situ synthesised oxidising/reducing agents, ensuring high material efficiency and circular use of non-hazardous chemicals.
- Mild emissions, with the possibility to further treat any effluents using conventional wastewater treatment methods.
- Low temperature operation: from room-to-mild temperatures [18 – 70 °C].
- Direct synthesis of functional battery materials, such as Co/Ni/Mn spinels and birnessites, which can be used to manufacture new lithium-ion battery electrode materials and eventually LIBs. This feature eliminates the need for additional refining steps, streamlining the production of new batteries from recycled material.
Within the RHINOCEROS project framework, researchers at VITO have been finetuning the operational parameters of their proprietary GDEx process, additionally testing and validating the recovered materials through the synthesis of high-performance electrodes for next generations batteries, demonstrating therefore the circularity, cost, environmental and social benefits of the solutions developed.
After testing the direct recovery route of Co, Ni, Mn and Li on synthetic solutions with the main objective to finetune the GDEx operating settings, VITO researchers have been implementing the process to the BM samples provided by the pre-treatment operations [ACC and TES] and to the aqueous extractants processed by the hydrometallurgical route explored by UoS and the solvometallurgical route explored by TEC. Additionally, they also documented the testing operations using a 3D phase diagram material library, mapping the formation of different materials based on Co/Ni/Mn concentration ratios from synthetic solutions.
The GDEx process applied on the leachate solution obtained from the BM provided by ACC achieved high recovery rates: 90 % for Ni, 89 % for Mn and 96 % for Co.
On the other hand, GDEx process implemented on the leachate solutions provided by UoS [originally from the BM of TES] results in the metal extraction efficiencies of 98 % for Ni, 68 % for Mn, and 96 % for Co. Sequential GDEx experiments have potential to improve the recovery efficiency by removing the impurities first and then the recovering the targeted metals. GDEx treatment on TEC’s leachate solution required optimisation due to the presence of high content of Cu and Al impurities.
With better results obtained on the lithiated nickel manganese cobalt oxide (LNMCO) material synthesised from the leached black mass provided by ACC [thermal pre-treatment], VITO researchers validated its electrochemical activity as a cathode material for LIBs with coin-type half cells. The recycled cathode material maintained a stable charge-discharge profile up to 50 cycles, with an initial charge-specific capacity of 37 mAh g⁻¹ and a reversible capacity of 26 mAh g⁻¹. Later, a batch with large amount of BM will be treated by GDEx process to optimise the electrochemical performance of the recycled electrode material.
After removing the targeted transition metals Ni, Co and Mn from the leachate solutions of the BMs, researchers used the same solution to extract lithium, recovering >99 % Li from the leachate of ACC BM.
The low-temperature or near-ambient operation condition of GDEx process eliminates CO₂ emissions, ensuring a clean and cost-effective approach to recovery of NMC precursor. This aligns with RHINOCEROS project goals, promoting a circular and sustainable battery materials economy. These results highlight the sustainability and scalability of the GDEx process, providing a carbon-free alternative for battery material production, aligning with global efforts to decarbonize critical raw material supply chains.
© visual: VITO
Authors: CHA and PNO
An important component of a Li-ion battery (LiB), the electrolyte has a crucial role in the cell performance. The nonaqueous electrolyte is a multicomponent system consisting of a conductive salt, mainly LiPF6 (Lithium hexafluorophosphate – inorganic component), organic carbonate solvents and additives.
Numerous research initiatives are dedicated to the design of the electrolyte composition, aiming to optimized performance, increased safety, lifetime and streamlined costs. However, once a LiB reaches its end-of-life (EoL) stages, the electrolyte receives less attention in the favour of the valuable metals that can be recycled from the cathode: Li, Co, Mn, Ni, Al, Cu. Moreover, due to its volatility, toxicity and high flammability, the electrolyte recycling is less studied. When ignored, the spent electrolyte reacts with water to form fluoride, leading to its uncontrolled decomposition and/or evaporation, and thus generating an imminent environmental risk. Due to its composition that includes organic solvents, the presence of residual electrolyte in the black mass is considered hazardous, and it is often a reason hampering the recycling process.
Researchers at Chalmers University (CHA) ) developed a sub- and supercritical carbon dioxide (sc-CO2) extraction process to selectively recover the electrolyte from spent LiBs. Sc-CO2 extraction technologies are already widely employed in the food, beverage, pharmaceutical, and cosmetic industry. In a nutshell, sc-CO2 forms when CO2 surpasses its critical point at 31°C and 73.8 bar pressure. In this so-called supercritical state, CO2 demonstrates optimal mass-transfer characteristics and can be fine-tuned to alter its physicochemical characteristics by adjusting pressure and/or temperature. However, CO2 proves ineffective as a solvent for high molecular weight polymers and highly polar ionic compounds. However, the addition of a co-solvent or a modifier can significantly improve the solubility properties of sc-CO2, making this alternative a suitable process to selectively extract LiPF6 after solvent removal.
Researchers at CHA investigated different process parameters (pressure, temperature, extraction times) to understand their impact on the extraction behavior of different electrolyte solvents such as ethylene carbonate. Their analysis included both qualitative and quantitative results indicating the composition and the purity. Simultaneously, the research also monitored the process exhaust stream for potential formation of LiPF6 decomposition products. The findings showed that that the non-polar electrolyte solvents like dimethyl carbonate, diethyl carbonate, and ethyl methyl carbonate were successfully extracted using low-density CO2. The more polar electrolyte components such as ethylene carbonate and propylene carbonate were stepwise extracted by gradually increasing the system’s pressure. The developed technology is a game changer not only for electrolyte recycling but also for increased workplace and transportation safety due to removal of the flammable and hazardous substances from battery black mass. The simplicity of the processing design is an additional advantage of the technology.
© Photo credit: Adobe
RHINOCEROS attending Shifting Economy Week
From 21 to 25 November 2023, the city of Brussels hosted the Shifting Economy Week, an annual event dedicated to showcasing transformative projects that aim to pave the way to an economy that is low-carbon, regenerative, and equally circular. The 2023 exhibitors’ line-up included, among other regional stakeholders, our partner Watt4Ever (W4E), industrial partner specialised in the development of innovative solutions for energy storage and management. W4E leveraged its presence at Shifting Economy Week to to raise awareness about the importance of circular economy principles in the context of the battery industry.
During the same event, W4E’s CEO, Aimilios Orfanos, was invited to speak at the BeCircular conference, an event dedicated to presenting concrete examples of circular economy approaches put in place by Brussels-based companies. He shared insights from W4E’s experience in developing second-life battery systems for electric vehicles, emphasising their potential benefits in terms of environmental impact and cost savings. Simultaneously, the CEO also highlighted the challenges faced by the industry in implementing circular business models, including regulatory barriers and market incentives.

RHINOCEROS at its second participation at Circular Wallonia Days
A few days after attending Shifting Economy Week, W4E represented the RHINOCEROS project at the Circular Wallonia Days, held on 13 and 14 December 2023. Centred around advancing the circularity of the batteries value chain, the event brought together stakeholders from academia, industry, and government to discuss strategies for improving the sustainability of battery production, use, and disposal. The focus topics covered recycling technologies, supply chain transparency, and policy measures to support the transition to a circular battery economy.
© Photo credits: Watt4Ever
Part of the Cluster Hub “Production of raw materials for batteries from European resources”, the RHINOCEROS consortium received the online visit of FREE4LIB representatives during the second day of the Consortium meeting held in Gothenburg. This initiative including stakeholders involved in different European R&D initiatives goes beyond building a knowledge exchange ecosystem to address common topics related to EU-funded projects; it paves new collaboration routes and synergies aiming at driving innovations for the recycling of batteries and the production of raw materials for battery applications from primary and secondary resources available in Europe.
Represented by Julius Ott (industrial engineer with expertise in circular economy at Karl-Franzens-Universität Graz) and Pau Sanchis (senior policy officer Eurobat), the FREE4LIB presentation focused mainly on the Digital Battery passport and the relevant legislative situation at European level.
Pau Sanchis referred to the Digital Battery Passport in the context of the new regulation on batteries and waste batteries which entered into force on 17 August 2023. According to this update, thoroughly explained in Art. 77, the battery passport should contain information “relating to the battery model and information specific to individual battery, including resulting from the use of that battery”.
“Batteries should be labelled in order to provide end-users with transparent, reliable and clear information about batteries and waste batteries. That information would enable end-users to make informed decisions when buying and discarding batteries and waste operators to appropriately treat waste batteries. Batteries should be labelled with all the necessary information concerning their main characteristics, including their capacity and the amount of certain hazardous substances present. To ensure the availability of information over time, that information should also be made available by means of QR codes which are printed or engraved on batteries or are affixed to the packaging and to the documents accompanying the battery and should respect the guidelines of ISO/IEC Standard 18004:2015. The QR code should give access to a battery’s product passport. Labels and QR codes should be accessible to persons with disabilities, in accordance with Directive (EU) 2019/882 of the European Parliament and of the Council (17).”
The policy officer emphasised the role of the standardisation process on the Battery Passport, which requires the Commission to adopt implementing decision requesting European Standardisation Organisation to develop standards in support of Ecodesign by December 2023. Standards regarding the technical design and operation of the Battery Passport are expected to complement provisions under Art. 78. According to the timeline presented in the regulation, the first application of the battery passport is expected in 2027. From 18 February 2027 onwards, “all batteries shall be marked with a QR code as described in Part C of Annex VI.
The new regulation aiming at strengthening sustainability rules for batteries and waste batteries will be supported by various secondary legislation pieces which will ensure all the requirements will be developed and implemented effectively. The QR code shall provide access to the following:
- for light means of transport (LMT) batteries, industrial batteries with a capacity greater than 2kWh and electric vehicles batteries, the battery passport in accordance with Article 77.
- for other batteries, the applicable information referred to in paragraphs 1 to 5 of this Article, the declaration of conformity referred to in Article 18, the report referred to in Article 52(3) and the information regarding the prevention and management of waste batteries laid down in Article 74(1), points (a) to (f).
- for starting, light, and ignition (SLI) batteries, the amount of cobalt, lead, lithium or nickel recovered from waste and present in active materials in the battery, calculated in accordance with Article 8.
The policy overview presentation was complemented by the technical presentation undertaken by Karl-Franzens-Universität Graz, that will develop a data model of the digital battery passport platform aiming to close the information gap between beginning-of-life (BoL) and end-of-life (EoL) battery lifetime. Relying on knowledge generated previously by Univ. of Graz, the researchers set an objective to define clear user roles and establish access to certain information. Up to this moment, the work carried out has been focusing on data collection and data handling (data points sorting) on the other side. This specific work encountered various challenges, notably the users willingness to share information, process standardisation, the variety of products, the recycling cost/revenue ratio, the dynamic development of the legislative framework, to name a few.
Learn more about the progress on the battery passport on the FREE4LIB website.
On Monday, 10 July 2023, the Council of the European Union adopted a new regulation that strengthens sustainability rules for batteries and waste batteries. This regulation covers the entire life cycle of batteries, ensuring their safety, sustainability and competitiveness from production to reuse and recycling.
Read the official press release
Recognising the vital role batteries play in the decarbonisation process and the transition towards zero-emission mobility, Teresa Ribera, Spanish Minister for the Ecological Transition reinforced the Presidency’s commitment to supporting comprehensive regulation encompassing all types of batteries. This includes waste portable batteries, electric vehicle batteries, industrial batteries, starting, lightning and ignition (SLI) batteries primarily used in vehicles and machinery, as well as batteries for light means of transport like electric bikes, e-mopeds, and e-scooters.
“At the same time end-of-life batteries contain many valuable resources and we must be able to reuse those critical raw materials instead of relying on third countries for supplies. The new rules will promote the competitiveness of European industry and ensure new batteries are sustainable and contribute to the green transition.”
| Teresa Ribera, Spanish Minister for the Ecological Transition
To foster a circular economy, the regulation establishes requirements for the end-of-life phase, including collection targets and obligations, material recovery targets, and extended producer responsibility. Dedicated collection objectives for waste batteries used in light means of transport will be implemented, aiming at 51% by the end of 2028, respectively 61% by the end of 2031. Furthermore, the regulation sets mandatory minimum levels of recycled content for industrial batteries, SLI batteries and electric vehicle batteries. The following initial values have been established:
- 16% for cobalt
- 85% for lead
- 6% for lithium
- 6% for nickel
Additionally, batteries will also be required to hold documentation proving their recycled content.
To improve the functioning of the internal market for batteries and ensure fair competition, the regulation introduces safety, sustainability, and labelling requirements. It includes provisions for battery labelling and information disclosure, including details on battery components and recycled content. Additionally, an electronic “battery passport” and a QR code will be implemented to enhance traceability and transparency. These labelling requirements will take effect by 2026, while the QR code implementation is expected by 2027, providing member states and manufacturers with ample time to prepare.
This new regulation aims to mitigate environmental and social impacts throughout the battery’s life cycle. By establishing strict due diligence rules for operators, the EU is ensuring operators are bound to verify the source of raw materials used for batteries placed on the market. However, the regulation provides for an exemption for SMEs from the due diligence rules.
After its signature by the Council and the European parliament, the new regulation will be published in the EU’s Official Journal, expecting to enter into force 20 days after.
RHINOCEROS project in the current legislative framework
Launched in 2022, the RHINOCEROS project fits within the current framework recently adopted by the Council of Europe under the Spanish Presidency. Designed to support the raw materials supply, the RHINOCEROS project will demonstrate a smart sorting and dismantling robot at TRL6, enabling the automation of a battery repurposing production line. When direct reuse and repurposing of batteries is not possible, RHINOCEROS will investigate several ground-breaking circular recycling routes aiming at the recovery of all materials present in LIBs (e.g., metals, graphite, fluorinated compounds, electrolytes, polymers, and active materials).
A first set of conclusions stemming from the research of our partners generated a database and the parameters for module selection, which will further facilitate the development of electric vehicles 2nd life batteries.
Read more in the article “Acceptance criteria and guidelines for 2nd life prone LIBs”
The infographic can be accessed on the Council of the European Union’s website using this link.
Aiming towards a zero-waste strategy for the recovery of metals from battery refining waste waters, LEITAT is working on the development and evaluation of novel polymer inclusion membranes (PIM). PIMs are a type of liquid membrane in which the liquid phase, the extractant, is held within a polymeric network. The interest in these membranes has been growing exponentially over the past few years as an alternative separation technique to conventional solvent extraction.
Work during the first six months has focused on the evaluation of different extractants for the target metals: lithium, manganese, cobalt and nickel. Researchers established a liquid-liquid extraction protocol based on two different processes in which the target metal is extracted and recovered separately. During the extraction step, a specific carrier compound selective towards the target metal separates an amount of it from a feed metal solution. The recovery of the metal takes place in the second process, where a stripping solution is employed to recover the metal previously extracted through the carrier. Initial PIMs containing the most efficient extractants have been prepared, characterised and are currently evaluated. The featured image depicts the continuous procedure used to test the synthesised PIMs.
The current industrial pre-treatment and downstream processes (e.g., pyrolysis, calcination, etc.) are still inefficient and have significant limitations. Plastics and electrolytes sacrificed in the initial stages of the recycling process are overlooked when it comes to their recovery. Within the RHINOCEROS project, Chalmers University [CHA] is working on recycling of ignored content of the LIBs waste, electrolyte and polymeric materials by developing an innovative process based on Supercritical Carbon Dioxide (sc-CO2) technology.
Due to its environmental friendliness, non-toxic, low cost and straightforward processing features, the sc-CO2 technology has been attracting both scientific and industrial interest. With consistent leverage over other processes, sc-CO2 technology is already widely used in various industries including food, cosmetic and pharmaceutical industries [i.e., to decaffeinate coffee or tea, extract vegetable oils etc.] Although it has many applications, its use in battery recycling was recently discovered and the Chalmers research group is one of the pioneers in this field.
Within the project’s framework, CHA researchers are targeting the development of the sc-CO2 extraction process which will selectively recycle the electrolyte and the polymeric material from the LiB waste. The electrolyte, binder, and separator will be recycled in subsequent steps and purified to reuse in the battery industry. For this purpose, several critical process parameters such as pressure and temperature are investigated to achieve high recycling efficiencies under feasible conditions.
Electrolyte recovery
The electrolyte in the LiB is a complex system usually composed of a conductive salt dissolved in a matrix of various solvents and additives. The most recent results reported by CHA on sub- and sc-CO2 research show that at low pressure and temperature conditions, the non-polar electrolyte components (almost 66% of the electrolyte) were selectively recovered without the generation of toxic gas emissions, which are typically generated by thermal recovery processes originated by the decomposition of the thermally unstable conductive salt.
In this recycling step, the polar electrolyte components are left in the sample as residues and a subsequent recycling step using suitable cosolvent is required for their selective extraction. During the upcoming phases, researchers will aim to recover the electrolyte completely, including conductive salt and solvents.
In this recycling step, the polar electrolyte components are left in the sample as residues and a subsequent recycling step using suitable cosolvent is required for their selective extraction. During the upcoming phases, researchers will aim to recover the electrolyte completely, including conductive salt and solvents.
The selection of the co-solvent is critically important not only for effective recycling but also for the sustainability and the feasibility of the developed processes. To assess the suitability of the sc-CO2 process, its environmental impact and economic competitiveness, CHA researchers explored also other solvents, which allowed them to select the most promising candidates for future research.
In the upcoming months, CHA will study the effects of co-solvent modified sc-CO2 system parameters on the recycling efficiency of PVDF and the structural properties of the recycled material. Researchers will carry out intensive characterisation studies to clarify the changes in the material structure, to determine the quality, and to optimise the process to reach the reusability goal.
